Advanced Casting Technologies/Chill Problem
Every engineer working in foundries has encountered the problem of chill at some point in their career. This problem, which occurs particularly in thin sections, is often associated with high cooling rates and/or insufficient inoculation. For this reason, foundries dealing with chill problems usually try to find a solution by increasing the amount of inoculant used or by experimenting with different inoculant materials. Although this may seem like the first solution that comes to mind, there may be a different underlying cause behind the formation of chill.
THERE MAY BE A DİFFERENT REASON FOR CHİLL PROBLEM THAN YOU THİNK.
This structure, which we call “chill” (English: chill), is actually a carbide structure known in metallurgy terminology as white cast iron. When conditions suitable for graphite decomposition are not provided during the eutectic transformation, the carbon dissolved in the liquid cannot separate as a separate phase in the form of graphite. As a natural consequence, carbon atoms remain dissolved in the liquid metal. During eutectic solidification, the carbon cannot be expelled from the liquid and solidifies, resulting in the hard and brittle structure we call carbide (chill).
When most foundries encounter the problem of chill, they may increase the amount of inoculant they use to solve the problem, based on the scenario described above. Although this may seem like the first solution that comes to mind, there may be a different underlying cause for the chill. To identify the true cause of chill formation and determine the correct solution, examining the microstructure of the formed carbide under a microscope often provides valuable insights.

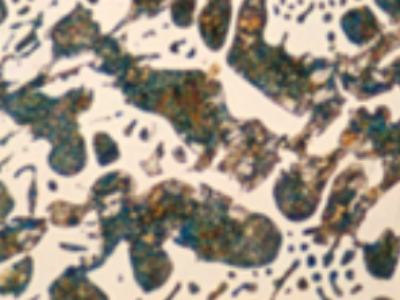
For example, if the formation of chill is really due to high cooling rates in thin sections or a problem related to overheating, then we see that the carbide has formed in a ledeburite structure (left image). When we examine this structure under a microscope, we see dark-colored islands within a continuous carbide phase that appears light in color.

Carbides formed in cast iron can also be seen under a microscope in the form of thin rods. These carbides, which actually precipitate in plate-like structures, appear as rods in a two-dimensional image taken through a cross-section. If the chill structure appears as rod-like carbides under a microscope, it can be concluded that too much Mg was used in the spheroidization process or that the amount of silicon, which is a graphitizing agent, was insufficient. Since magnesium is an element that tends to form compounds with both oxygen and sulfur, when an excessive amount of Mg is used, we observe that the oxygen and sulfur levels in the liquid drop to very low levels. As explained in various articles and lectures we have published on inoculation, a certain amount of oxygen and sulfur must be present in the liquid for the inoculant added to the liquid metal to function properly. When oxygen and sulfur levels drop to very low levels, graphite decomposition cannot be achieved, resulting in the inevitable formation of scale.
In the case of reverse chill, we see that the carbide particles exhibit a similar structure resembling a rod. This situation, which is observed mainly in large and circular sections, occurs due to the accumulation of carbide-forming elements in the liquid and their clustering in the middle section, resulting in a strong carbide-forming effect in this area.
Although chill is generally associated with rapid cooling and/or insufficient inoculation, we can see that this problem can also arise due to different causes. As explained above, a simple metallographic analysis allows us to identify the true cause of the problem and the correct solution. In other words, instead of relying on memorized methods, taking the time to examine the issue more closely enables us to easily identify the root cause and the appropriate solution.